Lipoproteins Tutorial
A Picture is Worth...
Physiology is complicated, complex, and constantly changing...it's hard to keep up with it. Trying to find a simple answer to a seemingly simple question becomes a challenge, especially when there are many conflicting hypotheses. Note-taking becomes tedious and boring, then trying to find 'that note' again becomes another chore. Try getting a large sheet of paper, colored pencils and a big eraser and draw a sketch of each answer you find. Keep them all on the same sheet...make it large...and your individual 'answers' will start fitting together into a single picture. And it's fun. Following is the result of this activity.
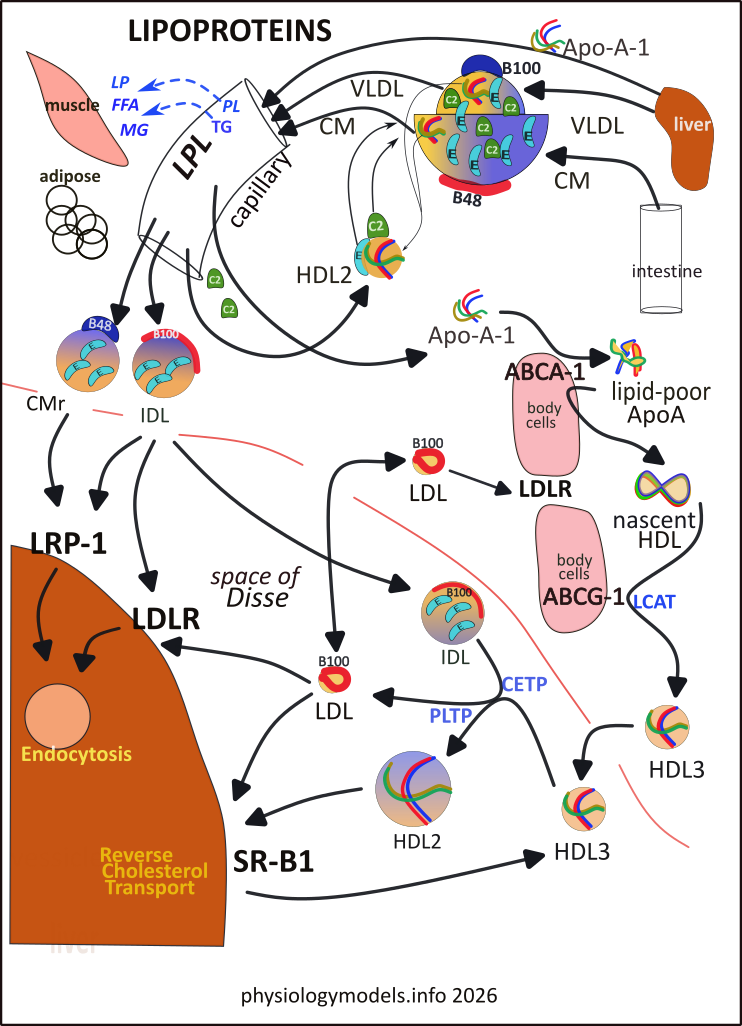
This illustration is the main 'physiology model'. The top third shows the 'birth' of both LDLs and HDLs with the liver producing VLDLs and Apo-A-1 particles and the intestine producing CMs. The 'grapefruit in a bowl' is a nested composite with the 'grapefruit' as the VLDL and the bowl as the CM...they're really spheres but the illustration shows half of each (the VLDL is really 1/10th the size of the CM).
The left half of the model shows these three particles flowing through capillaries where the VLDL and CM are partially drained of their contents becoming remnants (CMr and IDL). The Apo-A-1 particle penetrates the capillary wall...it is the 'baby' HDL. Follow it along the outer edge of the body cells. There it contacts several transporters that cause it to mature into an HDL3 particle.
The two remnants leave the circulation and enter the space of Disse around the liver. Here they encounter receptors that will endocytose them as vesicles. Escaping immediate 'distruction', IDL can interact with HDL3 (lower center) and, with the help of enzymes, becomes HDL2 and LDL.
LDL has several fates: (1) it contacts a liver receptor and is endocytosed, (2) it contacts a different receptor to have its contents 'sucked out', and (3) contacting body cells it is endocytosed or 'sucked dry' of its cholesterol.
HDL2, formed after receiving triglycerides from IDL, attaches to SR-B1 receptors that reduce it back to a smaller HDL3 that detaches and interacts with IDLs again.
***************************
Chylomicrons & VLDLs
Carefully observe the details of the 'bowl' in the model. Chylomicrons (CM), formed in the small intestine, consist of a single-layer coat of phospholipids with one permanent molecule of Apoprotein-B-100 (red), several semi-permanent Apo-A-1 proteins (multi-colored), and a core of ~90% triglycerides (blue). The amount of cholesterol (gold) depends on the diet.
Chylomicrons are absorbed into the lymphatic lacteals, (not illustrated). While there, they obtain copies of transient (detachable) Apo-E from HDLs in the lymph. They pass through the lacteals and into the thoracic duct that drains into the bloodstream.
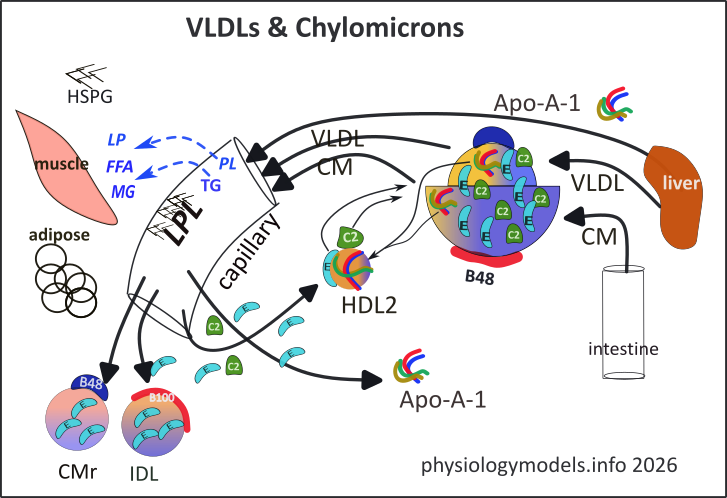
While in the circulatory system they transfer their Apo-A-1 molecules to HDL2 in exchange for transient Apo-C-II (green) and additional Apo-E (turquoise)...note thin arrows. This exchange also occurs between HDL2 and VLDL ('grapefruit').
The liver secretes very low density lipids (VLDL) and separate Apo-A-1 molecules (multicolored). The VLDLs are very similar to chylomicrons but 1/10th the size, on average. They display one large permanent molecule of apo-B-48 (dark blue) on their surface. Their core contains ~60% triglycerides and the rest is cholesterol esters (note colors). Their phospholipid coat also carries Apo-A-1 molecules.
On entering the circulation, both of these lipoproteins receive additional copies of the transient surface proteins Apo-C-II (green) and many Apo-E (turquoise) from HDL2. In exchange, the large lipoproteins transfer Apo-A-1 to HDL2 (note thin arrows). These transfers can happen by chance contact in the plasma but they can also occur while sequestered on Heparan Sulfate Proteoglycans (HSPG) bound to the capillary lining.
The large lipoproteins can't 'squeeze' through capillary walls but they do bind electrostatically to heparan sulfate proteoglycan (HSPG) attached to the endothelial lining. The Apo-C-II they carry activates the Lipoprotein Lipase (LPL), anchored to HSPG, that hydrolyzes their core triglycerides into free fatty acids and monoglycerides (MG); LPL also hydrolyzes phospholipids (PL) into fatty acids (FFA) and lyso-phospholipid (LP). These products are incorporated into surrounding tissues such as muscle and adipose.
With reduced surface area, many apoproteins they previously picked up from HDL2 (Apo-E & Apo-C-II) are shed into the plasma, but much Apo-E remains attached. The importance of Apo-E concentration on the remnants will be described in the 'Endocytosis' section.
The detachment of Apo-C-II causes the remnant to undock with LPL and the significantly smaller remnants are now free to circulate. The VLDL remnant is referred to as intermediate density lipoprotein (IDL) and the CM remnant is simply CMr.
The 'Endocytosis' section will follow the fate of the remnants. The fate of Apo-A-1 is described in the 'HDL Maturation' section.
***************************
Remnants
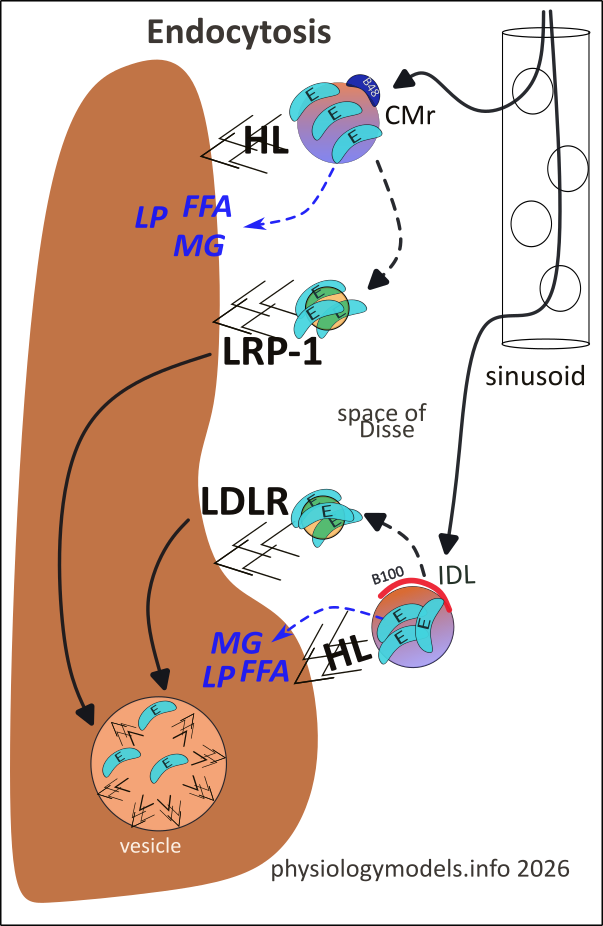
The capillaries in the liver are enlarged, porous (fenestrated) sinusoids. These fenestrae ("windows") allow the remnants to leave the circulation and enter a narrow space of Disse bathing the hepatocyte surfaces. The hepatocytes display many copies of negatively-charged Heparan Sulfate Proteoglycan (HSPG) that attract positively-charged Apo-E these remnants received from HDL2.
Hepatic Lipase (HL), sequestered by HSPG, hydrolyzes the adjacent remnant's triglycerides to free fatty acids (FFA) and monoglycerides (MG). This enzyme also hydrolyzes the remnant's surface phospholipids, to a free fatty acid and lyso-phospholipid (LP).
This activity continues shrinking the remnants causing Apo-Es to become more tightly packed together. It is the density of Apo-Es that enhances their chance of binding to receptors. Many copies of these receptors randomly diffuse through the hepatocyte cell membrane and come into contact with the sequestered remnants. The greater the Apo-E density the greater the probability of their binding to a nearby receptor.
There are two types of receptor: LRP-1 (LDL Receptor-related Protein 1) and LDLR (LDL Receptor). While sequestered, CMr components are hydrolyzed and the products (FFA, MG, LP) are taken into the hepatocyte (blue dashed arrow). The black dashed arrow illustrates its shrinken, Apo-E coated appearance and adjacent LRP-1. The arrow in the liver points to an endocytic vesicle where it is totally decomposed.
The IDL is processed in the same manner. The difference is that, after hydrolysis is sufficient to pack its Apo-Es, it is bound by LDLR. Regardless, the fate is the same...endocytosis.
***************************
HDL Maturation
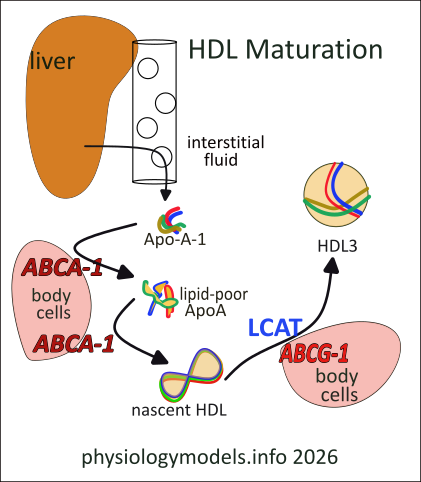
The liver secretes molecules of the Apo-A-1 into the space of Disse; at this point they are simply unorganized, coiled fibrous molecules (multi-colored). They enter the circulation by passing through the fenestrae in the hepatic sinusoids. Eventually they diffuse through capillary walls and into the interstitial fluid where they encounter cells throughout the body.
All body cells produce their own cholesterol as a structural component of their membranes. An excess of cholestrol in the cell membane signals the cell to produce ATP-binding cassette transporter A-1 (ABCA-1) on its surface. If Apo-A-1 particles contact this transporter, cholesterol and phospholipids are transferred to it; it is now referred to as lipid-poor Apo-A-1. Continued are re-contact allows additional transfers that convert this shapeless particle into disc-shaped nascent HDL.
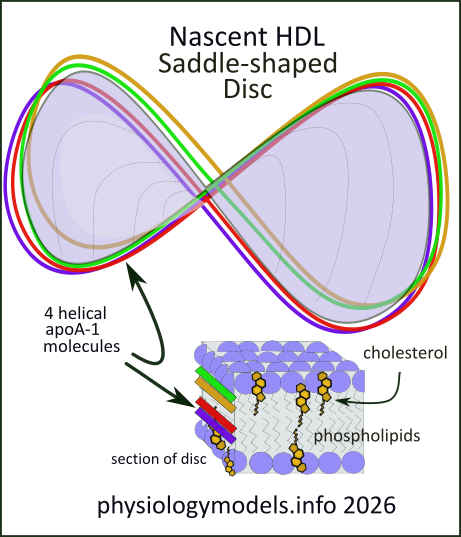
This conversion is detailed in this illustration. It shows how the phospholipid and cholesterol molecules self-assemble as they accumulate among the Apo-A-1 fibers. Cholesterol (C) and phospholipid (PL) molecules both elongated; they are hydrophobic (water-fearing) at one end and hydrophilic (water-loving) at the other. The cuboidal inset in the illustration shows how they assemble with the long hydrophobic fatty acid tails of phospholipids facing each other. This forms a double layer, with the phosphates (lavender balls) facing both surfaces.
There are four Apo-A-1 molecules; each is hydrophobic along one side and hydrophilic on the other. All four align their hydrophobic sides against the exposed hydrophobic 'edge' of the forming double membrane. They provide a girder that twists the final disc into the shape of a saddle. This structure is called nascent HDL.
Still in the interstitial fluid, nascent HDL is recognized by another similar transporter, ABCG-1. This also transports cholesterol and phospholipids that, with the help of the plasma enzyme Lecithin-Cholesterol Acyltransferase (LCAT) converts the free cholesterol into cholesterol ester (CE). This compound is completely hydrophobic and pushes between the two layers to form a sphere. The encircling Apo-A-1 proteins change their orientation and continue to provide scaffolding for the sphere. This structure is referred to as HDL3. Observe it in the left illustration.
***************************
Lipoprotein Interactions
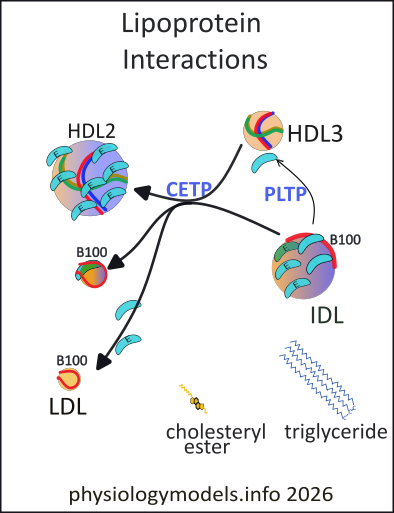
Cholesterol ester transfer protein (CETP) is a plasma transporter that serves as a bridge between HDL3 and IDL. It forms a tunnel between them allowing the two-way diffusion of cholesterol esters and triglycerides. The result is that the larger IDL loses triglycerides to the smaller HDL3 particle in exchange for its cholesterol esters. Triglyceride molecules are larger than cholesterol esters (as illustrated) and the exchange reduces the size of the IDL and increases the size of the HDL3; it becomes HDL2.
Wondering why HDL2 is larger than HDL3? The numbers do not represent the size of the particle. They represent their relative densities. The amount of heavy Apo-A-1 is the same on each, but there are more per unit volume on the smaller particle...it is the more dense of the two.
The color gradients indicate the approximate percentages of TG (blue) and CE (gold). Initially, HDL3 is 100% CE while IDL is ~ 50:50. In the products, these values are changed to HDL2 ~60% TG and 40% CE while the LDL may have a small amount of TG remaining. Otherwise it will be a Apo-E free LDL.
Shrinkage of IDL displaces excess membrane phospholipids and transient proteins. The plasma transporter phospholipid transfer protein (PLTP) transfers them to growing HDL particle (thin arrow). As IDL continues to shrink it can become so small that it sheds all its Apo-E proteins forming the 'infamous' LDL.
***************************
Cholesterol Transport
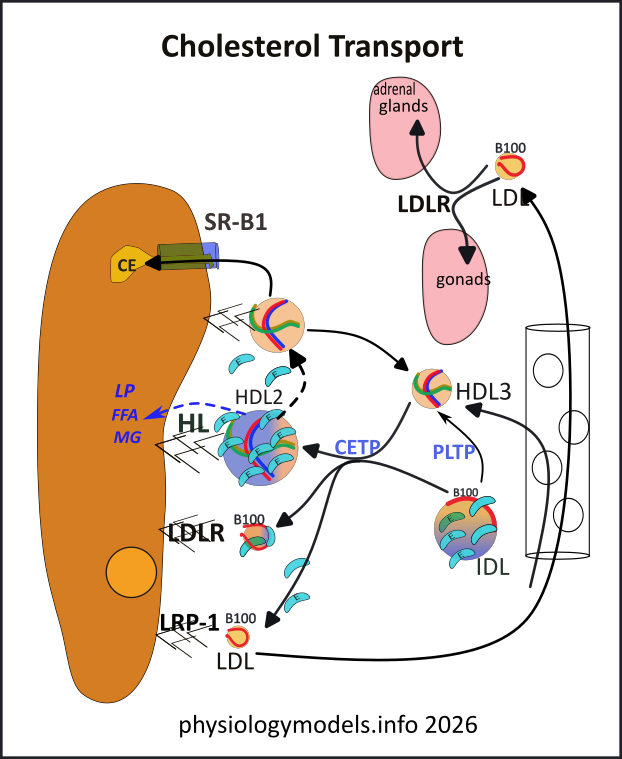
LDL, formed in the space of Disse, is a vital source of cholesterol for specific tissues. Leaving that space, it circulates to squeeze between capillary endothelial cells to find its targets. The adrenal glands require massive amounts of it for the production of the steroidal hormones cortisol, aldosterone, and androgens. The liver and gonads also require cholesterol for the production of bile and sex hormones.The entire LDL particle is endocytosed providing abundant cholesterol.
HDL particles have been maturing and growing as they accumulated cholesterol esters from cholesterol-heavy body cells..recall their activities in the interstitial fluid involving the ABC transporters and LCAT. The 'mature' HDL3 returns to circulation and enters the space of Disse. There it gains TG and loses CE to IDL becoming HDL2.
At the liver's surface the HDL2 triglycerides and phospholipids are hydrolyzed by HL held in HSPGs. As the particle shrinks it loses it transient apoproteins and its unmasked Apo-A-1 becomes linked to Scavenger Receptor B-1 (SR-B1). This receptor forms a tunnel through which CE diffuses until an equilibrium is reached. The still-shrinking HDL eventually becomes too small to remain attached to SR-B1 and detaches. Its relative high proportion of heavy Apo-A-1 reclassifies it as HDL3.
HDL2s delivery of CE is only part of the cholesterol in the original HDL3; the other portion is now in LDL. The LDL, shrunken to the point where Apo-E proteins have been shed, is endocytosed by LDLR or LRP-1 thus delivering the final cholesterol load to the liver. Another route that LDL could also travel is to the body cells that display LDLR, such as the adrenal glands and gonads.
***************************
A Thousand Words
VLDLs and Chylomicrons
The liver produces apoprotein A-1 (Apo-A-1) and very low density lipoproteins (VLDL). Apo-A-1 is released as four entangled coiled strands of protein. VLDL is a spherical lipid particle with a single-layer phospholipid coat and a core of ~55-65% triglyderides and ~10-15% cholesterol esters. There is a single, permanent Apo-B-100 protein embedded in its coat in addition to several Apo-A-1 proteins.
The small intestine produces chylomicrons (CM). They are 10X larger than VLDLs. Their coat is the same as VLDL with a single apoprotein B-48 (Apo-B-48) embedded. It also carries copies of Apo-A-1. It contains ~85-92% triglycerides and ~1-3% cholesterol esters...depending on the diet.
On entering circulation they shed their Apo-A-1 proteins to high density lipoprotein 2 (HDL2) in exchange for copies of Apo-C-II and Apo-E. These are transient proteins that are readily transferred between lipoproteins. With their permanent B apoprotein and transient Apo-C-IIs and Apo-Es these large lipoproteins become sequestered on heparan sulfate proteoglycans (HSPG) lining the capillaries. These molecules are negatively charged and the positively charged Apo-E proteins are attracted to them.
The Apo-C-II the remnants carry activates lipoprotein lipase (LPL) on the endothelium causing their triglycerides to hydrolyze to free fatty acids (FFA) and monoglycerides (MG); their phospholipids hydrolyze to FFA and lyso-phospholipid (LP). The products are taken into the tissues the capillaries pass through. The cholesterol esters are not affected. As a result, the remnants are approximately half TG and half CE.
As they shrink, the excess coat material is shed along with the transient apoproteins. Loss of Apo-C-II breaks the connection with LPL and the remaining lipoprotein (remnant) is released into circulation. The shed Apo-C-II and Apo-A-1 but not Apo-E. They shed proteins are picked up by HDL2 to participate in another exchange with VLDLs and CMs.
Remnants
The remnants, intermediate density lipoprotein (IDL) and chylomicron remnant (CMr), pass through the fenestrae of liver sinusoids to enter the space of Disse. Again, they are sequestered by HSPG. In the liver, the enzyme Hepatic Lipase (HL), also held by HSPG, hydrolyzes their triglycerides and phospholipids...reminiscent of the prior capillary activity. Again, the products are taken up by the cells.
There are two types of receptors that will bind the smaller, hydrolyzed remnants. The LDL Receptor (LDLR) preferentially binds with what's left of IDL while LDL Receptor-Related Protein 1 (LRP-1) prefers the shrunken CMr. Their attachment to the receptors depends on the remnants being densely covered with Apo-Es. This occurs naturally as the remnants shrink...Apo-E is not that easily shed. With the cell surface covered with these small, bound, cholesterol-rich particles it invaginates and forms an endocytotic vesicles. Internally, the contents of a vesicle are hydrolyzed; the HSPG, Apo-E proteins and vesicle membrane are trafficked back to the surface. And so it goes...
HDL
The Apo-A-1 the liver released is a group of four coiled proteins; the grouping has no particular shape. It enters the interstitial fluid through small gaps between endothelial cells lining capillaries. All body cells produce their own cholesterol for their membranes but, if in excess, they display the ATP-binding Cassette Transporters; there are two types, ABCA-1 and ABCG-1. On the other hand, if cells are in need of cholesterol the down-regulate these transporters and display LDLR receptors.
On contacting ABCA-1 transporters on cholesterol-rich cells, free cholesterol and phospholipids from the cell membrane are transferred to Apo-A-1. This forms lipid-poor ApoA. Continued attachment allows more additions and a double-layered membrane self-assembles within an Apo-A-1 girder. It forms a disc that is twisted into a saddle shape. It is called nascent HDL.
Having formed this shape, it detaches from ABCA-1. However, ABCG-1 does bind with this shape and also spherical HDL particles. This transporter continues supplying free cholesterol and phospholipids and now the plasma enzyme Lecithin-Cholesterol Acyltransferase (LCAT) converts the free cholesterol into cholesterol ester that is entirely hydrophobic and assembles between the two phospholipid layers of the disc...and increases the volume of spherical particles. The small, spherical HDL formed is HDL3. Its Apo-A-1 proteins form an encircling scaffolding to give it support. Additional Apo-A-1 proteins are added as the sphere continues to grow.
Interactions
HDL3 leaves the interstitial fluid and eventually ends up in the space of Disse...where it will end up being continually recycled. There are free-floating Apo-Es in the space that attach to it and bind it to HSPG. If adjacent to a bound IDL the plasma transporter Cholesteryl Ester Transfer Protein (CETP) forms a tunnel between them. This allows the two-way diffusion of triglycerides (from IDL to HDL3) and cholesteryl esters (from HDL3 to IDL).
The HDL2s volume increases significantly on receiving large triglyceride molecules from IDL that shrinks significantly. The once boyant IDL is left a shadow of its former self; it is now a small, cholesterol-rich LDL. The LDL attaches to receptors on the liver, adrenal glands, and gonads to be endocytosed and supply cholesterol to these organs.
The enlarged HDL, now called HDL2, loses its newly-acquired triglycerides via hepatic lipase (HL) activity at the hepatocyte surface. As it shrinks it becomes cholesterol-rich again and, losing its Apo-Es, attaches to Scavenger Receptor B-1 that drains its cholesteryl esters into the hepatocyte. Continued shrinkage reduces its size to the extent that it detaches from the receptor. Free in the space it reacquires Apo-Es and interacts with IDL again.
***************************
Atherosclerosis
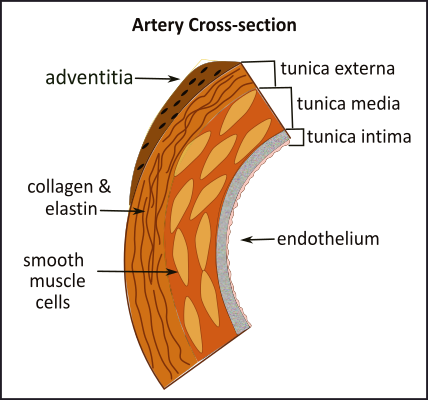
Places where arteries bifurcate such as the internal and external carotids, and where there are sharp curves (aortic arch) experience shear stress from rapidly flowing blood. Over time, this affects the endothelial cells that become damaged and allow impacting LDL particles to enter the intima.
This illustration is a simple reminder of the histology of an artery. Atherosclerosis is the slow buildup of a dense arthrosclerotic plaque...a 'scab' over a region of damaged endothelium. Most of the 'activity' occurs in the tunica intima; these will be shown in the following illustrations.
Foam Cell Formation
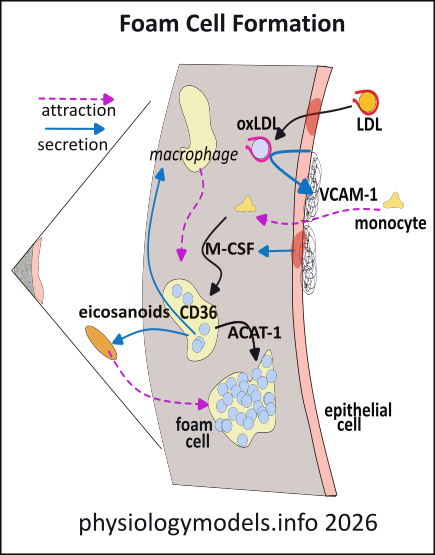
This enlargement of a small section of the tunica intima that shows an area of damaged endothelium (dark spot). This region allows LDL particles to enter the intima where oxidizing agents 'steal' electrons from their phospholipid coat and Apo-B-100 thus oxidizing the LDL. (Oxidation is the loss of electrons.) Being removed from circulation, LDLs can no longer access antioxidants...that replace stolen electrons...such as plasma Vitamin E.
The oxidized LDLs (oxLDL) activate endothelial cells to form VCAM-1 (blue arrow) adhesion molecules at their luminal surface . Circulating monocytes are attracted (pink arrow) there and squeeze through the endothelium into the intima. There they are stimulated to differentiate into macrophages by Macrophage Colony-Stimulating Factor (M-CSF) secreted by the damaged endothelial cells.
Macrophages display CD36 receptors that bind to oxLDLs and endocytose them. Within the macrophage, lysosomes decompose the LDLs cholesteryl esters to free cholesterol and a fatty acid. Most of these molecules are re-esterified by the enzyme ACAT1 and form 'greasy globs' changing the macrophages into foam cells. However, certain fatty acids are converted into eicosanoids (signaling lipid molecules) that recruit additional macrophages.
The signaling molecules also cause smooth muscle cells from the arterial wall to migrate into the intima. Once there, they also endocytose oxLDL and produce 'greasy globs' of lipids to also become foam cells. With the passage of time this region slowly grows producing a 'fatty streak' just below the surface of the artery.
Cap Formation
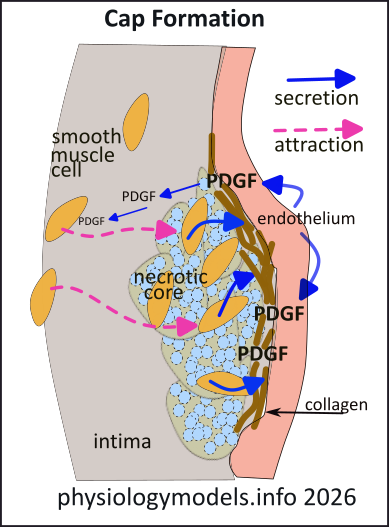
The foam cells become overwhelmed by lipid accumulation and die. Since they cannot be cleared from this region they leave a 'necrotic core' of extracellular lipid and cellular debris. The endothelial cells now begin secreting Platelet-Derived Growth Factor (PDGF) (blue arrows) that diffuses into the intima. Smooth muscle cells follow the gradient (pink arrow) of this chemical to the surface of the 'necrotic core'. Here they secrete collagen and elastin (brown) that form a 'fibrous cap' over the region.
If collagen comes into contact with clotting factors in the blood a thrombus (clot) will form. HSPGs attached to the luminal surface of the endothelium repel clotting factors to help prevent this.
Plaque Formation
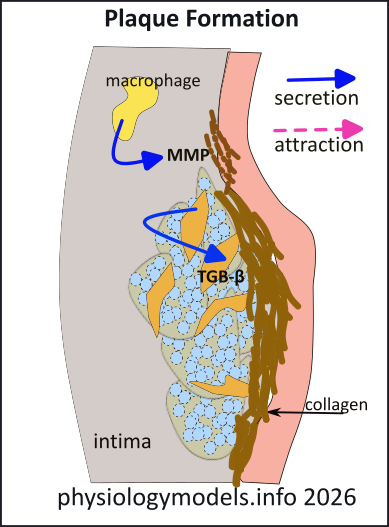
Still-viable macrophages in the core secrete Matrix Metalloproteinases (MMPs) that break down collagen. They also secete cytokines (signaling protein molecules) that inhibit the cap-building smooth muscle cells. The thinning, brittle cap of this 'hardened artery' is prime for rupture.
Any hemodynamic event such as a spike in blood pressure or heart rate might be sufficient to 'peel up' the edge of the plaque. This exposes the collagen, initiating a thrombus (blood clot) that extends into the lumen. If a piece of the thrombus (embolus) breaks off and enters a coronary artery the result is a heart attack, if it enters the brain the result is a stroke.
***************************
Appendix
- Enzymes
- LCAT (Lecithin-Cholesterol Acyltransferase)
- LPL (Lipoprotein Lipase)
- HL (Hepatic Lipase)
- ACAT-1 (Acetyl-CoA Acetyltransferase 1)
- MMPs (Matrix Metalloproteinases)
- Membane Transporters
- ABCA-1 (ATP-Binding Cassette Transporter A1)
- ABCG-1 (ATP-Binding Cassette Transporter G1)
- Plasma Transporters
- CETP (Cholesteryl Ester Transfer Protein)
- PLTP (Plasma Phospholipid Transfer Protein)
- Receptors
- LRP-1 (LDL Receptor-related Protein 1)
- LDLR (LDL Receptor)
- SR-B1 (Scavenger Receptor Class B Type 1)
- CD36 (Cluster of Differentiation 36)
- SR-A (Class A Scavenger Receptor)
- Cell Surface Anchors
- HSPG (Heparan Sulfate Proteoglycan)
- VCAM-1 (Vascular Cell Adhesion Molecule-1)
- Apoproteins
- Apo-A-1
- ApoE
- ApoC-II
- ApoB-100
- ApoB-48
- Molecular Mediators (cytokines and eicosanoids)
- PDGF (Platelet-Derived Growth Factor)
- TGF-ß (Transforming Growth Factor-beta)
- MMP (Matrix Metalloproteinase)
- MC-SF (Macrophage Colony-Stimulating Factor)
- Reactions
- cholesterol + lecithin → cholesterol ester + lyso-lecithin (enzyme LCAT)
- phospholipid → lyso-phospholipid + fatty acid (HL)
- triglyceride → 2 free fatty acids + monoglyceride (LPL, HL)